Incorporating Dental Sleep Medicine Into Your Practice
Snoring and obstructive sleep apnea (OSA) are common conditions that dentists can screen for and effectively treat with a custom-made mandibular advancement device (MAD). As dentists look to expand the services offered by their practice and contribute to their patients’ overall health, dental sleep medicine has emerged as one of the fastest-growing areas of dentistry.
For over 25 years, Glidewell has provided dentists with oral appliances for the treatment of snoring and OSA. More recently, we’ve initiated the provisional mandibular advancement device (PMAD) therapy protocol, in which dentists can easily identify patients who complain of daytime sleepiness or bed partner disturbance due to untreated snoring. Dentists can then offer single-visit sleep treatment using a provisional diagnosis along with our proprietary informed consent form, seeing to the patient’s primary complaint before referral to a physician for follow-up and medical management.
Dental Sleep Treatment Requires a Medical Diagnosis. Or Does It?
The provisional mandibular advancement device (PMAD) protocol offers dentists a means to simply and reliably treat a vast patient population suffering from snoring and obstructive sleep apnea.
PMAD Protocol: Sleep Therapy in 3 Easy Steps
One in four patients in the dental practice suffers from sleep-disordered breathing (SDB), which includes snoring and obstructive sleep apnea.1 The resulting medical conditions can include daytime sleepiness, diabetes, heart disease and stroke. For dentists starting out in sleep therapy, these are not new patients that have to be recruited, but existing patients who are easily identified if you ask the right questions.
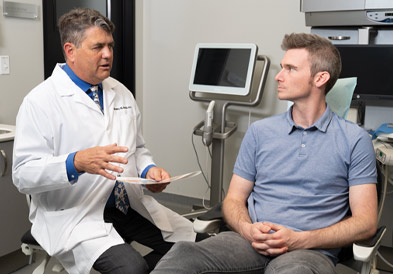
Step 1: Identify and Qualify Patients for PMAD Therapy
Step 2: Informed Consent
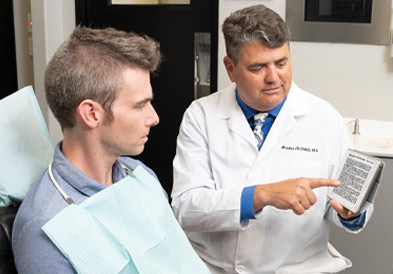

Step 3: Provide Provisional Treatment
- Move the jaw forward to activate muscles and ligaments of the oropharynx
- Provide more room for the tongue by separating the teeth and allowing the tongue to naturally posture forward
- Maintain an advanced jaw position during sleep to hold the tongue out of the airway

Silent Nite® 3D
- PDAC (Pricing, Data Analysis and Coding) contractor-verified: Code K1027 (for private insurance*)
- Precision fit due to fully digital workflow†
- Designed with more tongue space for enhanced comfort
- Each case includes extra connectors and a free AM Aligner®

Silent Nite
- PDAC (Pricing, Data Analysis and Coding) contractor-verified for Medicare reimbursement: Code E0486
- More than 400,000 Silent Nite Sleep Appliances delivered
- Metal-free and adjustable with up to 6 mm of advancement
- Each case includes extra connectors and a free AM Aligner
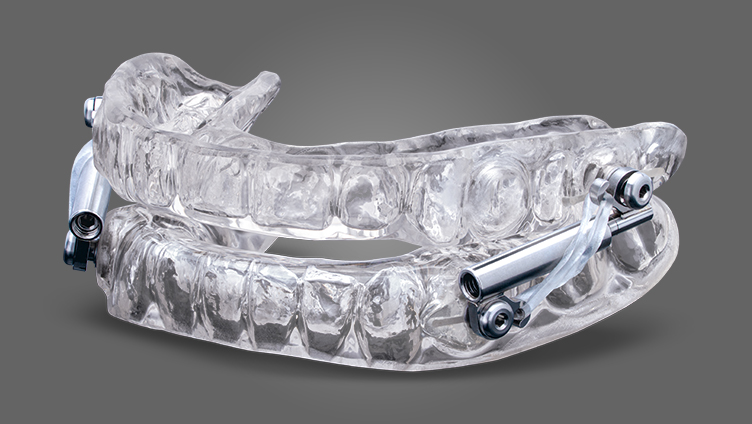
Silent Nite with
Glidewell Hinge
- PDAC (Pricing, Data Analysis and Coding) contractor-verified for Medicare reimbursement: Code E0486
- Micro-adjustable, with up to 10 mm of advancement
- Features telescope-style hinge
- Each case includes a free AM Aligner
For any of the above oral appliances, take a bite registration with upper and lower impressions. Submit these with your Rx form to Glidewell.
Payment for PMAD Therapy
Dental insurance does not pay for sleep appliances. Medical insurance can be complex and time-intensive, and will not pay unless a physician provides the definitive diagnosis. Therefore, the patient is charged directly for the provisional appliance and related chairside services. With affordability in mind, Glidewell offers sleep apnea treatments at the lower end of the cost spectrum. The popular Silent Nite Sleep Appliance is a treatment that can be fabricated for our most affordable price. Special pricing enables a backup appliance to be prescribed at the same time for greater savings.
Resources
1. Heinzer R, Vat S, Marques-Vidal P, et al. Prevalence of sleep-disordered breathing in the general population: the HypnoLaus study. Lancet Respir Med. 2015 Apr;3(4):310-8.
*Covered by many private medical insurance carriers but not covered by Medicare; check with your patient’s insurance policy to determine eligibility.
†All impressions must be submitted digitally to ensure maximum accuracy and speed; prescriptions via traditional impressions will be filled with a thermoformed Silent Nite appliance.